Candy Experiments (aka What to do with ALL that Halloween candy?!?)
So … what to do with ALL that Halloween candy? I definitely don’t want us eating all of it … therefore, in the name of science we will sacrifice some!
This worked especially well because Henry’s awesome kindergarten teacher (Mrs. Walch) sends home tons of great “homework” activities, one of them being the “Scientist of the Week bag”. This bag contains a labcoat, safety goggles, a book of science experiments (“Apples, Bubbles, and Crystals: Your Science ABCs” by Andrea Bennett & James H. Kessler), and a note explaining the assignment. Kids and parents are allowed to use any appropriate science experiment, not just the ones in the book. And, great timing, this week Henry got to bring the bag home and I saw the link to Candy Experiments … hence a wonderful morning of trying out different candy experiments to find the one to bring to class. (it did take some convincing Henry that using some of his candy for this, instead of eating it … but once I did we had a great time together!)
Acid Test
 A simple experiment to test whether or not candy has acid in it. As recommended we used sour candies (sour Skittles to be exact), water, and baking soda. If the candy has acid, the mixture will bubble/fizz when the baking soda is added.
A simple experiment to test whether or not candy has acid in it. As recommended we used sour candies (sour Skittles to be exact), water, and baking soda. If the candy has acid, the mixture will bubble/fizz when the baking soda is added.
Sink or Float?
We tested whether or not candy would sink or float to discover that all but 3 Musketeers and Whoppers sink. It was great to watch Henry think about why this would be so … he thought about how bubbles float because they are filled with air … sooo, the candy that floats must have more air in them! LOVE IT.
Stickyness
 Using our Laffy Taffy we found what makes it stickier … water, warmth, and playing with it in our hands. We then molded into a little sticky car!
Using our Laffy Taffy we found what makes it stickier … water, warmth, and playing with it in our hands. We then molded into a little sticky car!
Floating M’s
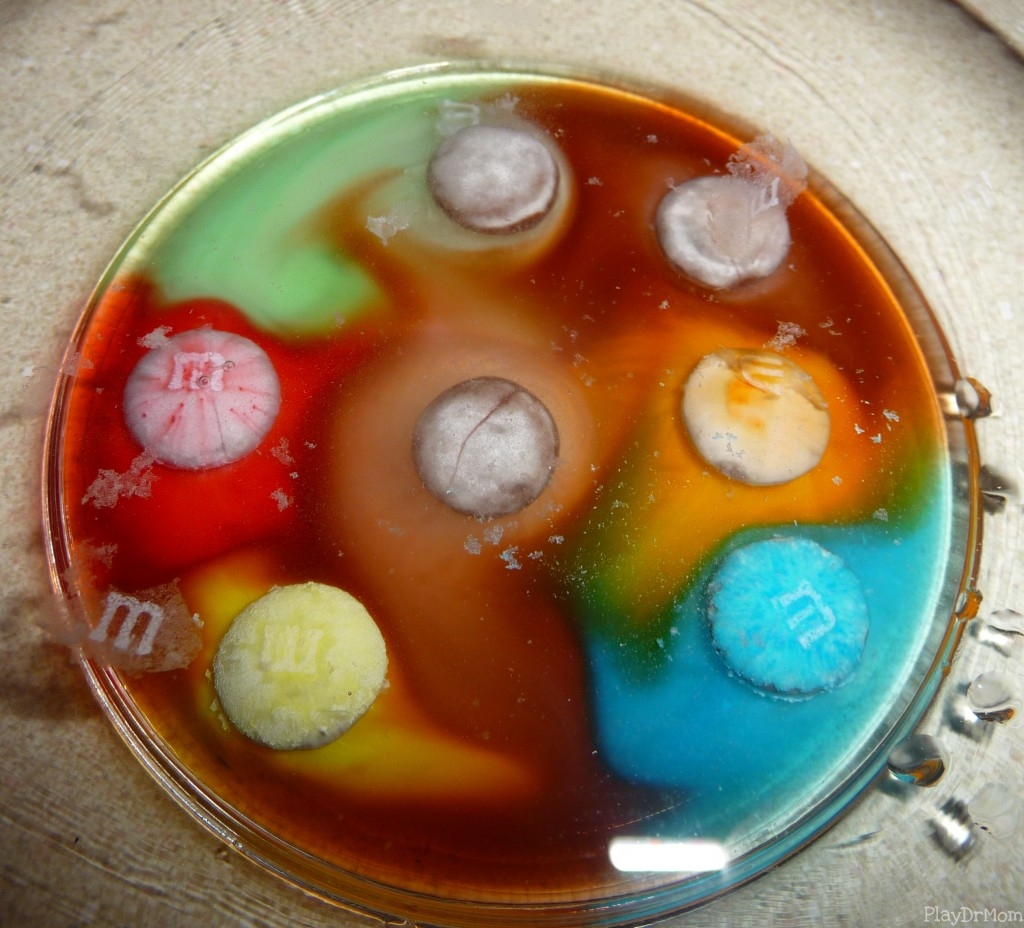
Using M&Ms and warm water we dissolved the candy and watched the m’s float to the top (because the edible ink used does not dissolve in water). This ended up being our favorite by far … and Henry will be demonstrating this experiment to his kindergarten class tomorrow!
Laura Hutchison
Latest posts by Laura Hutchison (see all)
- Hacked By GeNErAL - August 21, 2015
- ABCs of Easy Crafts for Kids - August 14, 2015
- What is Learned through PLAY - August 12, 2015
- ABCs of Playground Parkbench - August 7, 2015
- ABCs of Encourage Play - July 31, 2015





















Thanks for sharing this at Mom’s Library. Love all the activities – a fun way for the kids to learn some science concepts, better than use those chemical stuff. Featuring it on iGameMom.
iGameMom recently posted…Creative Learning Activities with Candy